
Chris Mantsounga, PhD
Brown University Health & Vascular Research Laboratory at the Providence VA Medical Center
830 Chalkstone Avenue
Building 35
Dr. Chris Mantsounga is a Research Biologist at the Ocean State Research Institute and a Research Assistant Professor of Medicine at the Warren Alpert Medical School of Brown University. He earned his Ph.D. in Cardiovascular Pathology from Paris Descartes University, following master’s degrees in Blood Vessels and Hemostasis, Genomes and Biology, and Cell Biology from universities in France. Dr. Mantsounga’s research focuses on the molecular mechanisms regulating inflammatory angiogenesis and vascular repair in aging and diabetes. His current projects, supported by the American Heart Association and NIH, investigate the roles of VEGF-A isoforms, macrophage signaling, and posttranscriptional regulation in impaired vascular regeneration. His work has been published in high-impact journals such as Cell Reports, Arteriosclerosis, Thrombosis, and Vascular Biology, and Biomedicines.
Diabetes mellitus (DM) is a chronic disease and a major health problem worldwide, with devastating consequences on morbidity, mortality and quality of life1-3. Patients with DM experience vascular/cardiovascular manifestations which include the peripheral artery disease (PAD). DM increases the risk of developing PAD while PAD contributes to the progression of DM4,5-. Critical limb ischemia (CLI), a severe complication of PAD-mediated diabetes mellitus (PAD-DM) is associated with failure blood supply and reduced neovascularization to the lower limbs. This is aggravated with age (>65 years) and can result in limb amputation or immobility6-8. Current treatment options are largely limited to mechanical revascularization, by surgical bypass or angioplasty. Also, medical management in PAD-DM may include anticoagulants, antiplatelet therapy, infection control and local ulcer care in DM patients 9,10. However, not all patients are eligible for surgery and angioplasty. Therapeutic angiogenesis strategy in PAD-DM is an attractive concept. However, preclinical and clinical gene-based therapies using angiogenic molecules like the vascular endothelial growth factor A (VEGF-A) failed to restore complete functional vascular networks11,12. CLI and wound injury animal models have highlighted the impact of monocytes/macrophages as a major source of angiogenic mediators13-16. We have recently defined a key mechanism whereby macrophage IL-1β promotes VEGF-A expression under the regulation of transcription factors in young mice13. Type 2 experimental diabetic model (T2DM), leptin receptor (Leprdb/db) mice at 10-weeks of age demonstrated delayed blood flow recovery compared to control, using a PAD model of femoral artery ligation that involves macrophage-directed blood flow recovery. “Aged” 52-week-old control mice also showed reductions in blood flow recovery. Moreover, combining aging with long-term diabetes, 52-week-old T2DM mice, led to further reductions in blood flow recovery consequent to impaired angio/arteriogenesis. RNA sequencing data from bone-marrow-derived macrophages (BMDMs) and single nucleus RNA sequencing from ischemic muscle tissue from “aged” T2DM mice, showed an exacerbation of Cxcl2 (MIP-2: macrophage inflammatory protein-2)-NLRP3-IL-1β pathway while VEGF-A expression and new blood vessel growth were not fully recovered compared to control in response to vascular injury. Aged T2DM mice also showed reductions in expression of myeloid differentiation primary response protein 88 (MyD88). Here, we aim to define the molecular mechanisms of reduced inflammatory angiogenesis by investigating the Cxcl2-NLRP3-IL-1β pathway in the context of chronic DM. T2DM mice were allowed to age and Cxcl2 neutralizing antibody will allow us to define the direct impact on the NLRP3-IL-1β-independent MyD88 expression. Hypothesis: PAD-mediated diabetes mellitus is associated with impairment of inflammatory angiogenesis related to exacerbated and dysregulated inflammatory response causing ineffective pro-regenerative response to vascular injury
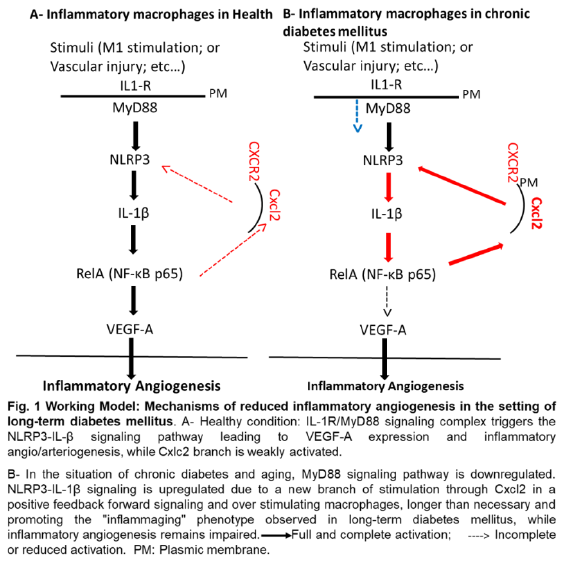
(Fig. 1 working model)
Autocrine Cxcl2-NLRP3 inflammasome activation as a mechanism of impaired inflammatory angiogenesis in the setting of chronic diabetes mellitus
Ocean State Research Institute
 Providence VA Medical Center
Providence VA Medical Center
Building 35
830 Chalkstone Avenue
Providence RI 02908
T: 401-273-7100
Research Funded by
Research reported in this website was supported by the National Institute of General Medical Science of the National Institutes of Health under grant number P20GM103652.




